Can Traumatic Memories Be Rewritten?
Preliminary TAU findings suggest noninvasive brain stimulation may reduce intrusive PTSD symptoms
A new study conducted at Tel Aviv University introduces an innovative approach to treating post-traumatic stress disorder (PTSD), generating particular interest in light of the sharp rise in the number of individuals coping with the condition following the events of October 7 and the Iron Swords War. According to the study’s preliminary findings, treatment using noninvasive brain stimulation succeeded in significantly reducing intrusive memories, such as flashbacks and intrusive thoughts, which are considered among the most severe and treatment-resistant symptoms of PTSD.
The study was conducted in the laboratories of Prof. Nitzan Censor and Yair Bar-Haim from the School of Psychological Sciences and the Sagol School of Neuroscience at Tel Aviv University. It was led by doctoral students Or Dezachyo and Noga Yair, in collaboration with the laboratory of Prof. Ido Tavor. The research team included Noga Mendelovitch, Dr. Niv Tik, Dr. Haggai Sharon of Tel Aviv Sourasky Medical Center (Ichilov), and Prof. Daniel Pine of the National Institute of Mental Health (NIMH) in the United States. The study was published in the scientific journal Brain Stimulation.

Research team (Left to right): Prof. Yair Bar-Haim, Noga Yair, Or Dezachyo and Prof. Nitzan Censor
A New Approach to PTSD Treatment
PTSD affects millions of people worldwide, including soldiers and survivors of terrorist attacks, traffic accidents, and violence. Despite advances in psychological and pharmacological treatments, only about 50% of patients respond well to existing therapies, and intrusive memories continue to burden many of them years after the traumatic event. These memories are not just distressing thoughts; they are vivid, tangible experiences that reactivate the body and emotions as though the trauma were happening all over again.
The researchers focused on the hippocampus — a deep brain structure responsible for the processing, storage, and retrieval of memories. Because direct stimulation of deep brain regions requires invasive intervention, the team employed an indirect and sophisticated method: they identified superficial brain regions that are functionally connected to the hippocampus and stimulated them using transcranial magnetic stimulation (TMS). The precise stimulation site was determined individually for each participant based on fMRI scans, allowing for a personalized treatment approach.
Promising Early Results
Ten adults with PTSD participated in the initial study, undergoing five weekly treatment sessions. During each session, the traumatic memory was first deliberately reactivated, after which brain stimulation was applied — precisely at the stage when the memory is in a “flexible” state and more open to change, within a process known as reconsolidation. The researchers’ aim was to influence the way the memory is re-stored in the brain, thereby alleviating post-traumatic symptoms.
The results showed a sharp reduction in the severity of post-traumatic symptoms, particularly in the frequency and intensity of intrusive memories, with participants demonstrating consistent improvement. At the same time, brain imaging revealed reduced connectivity between the hippocampus and the stimulation regions — evidence that the effects were not merely subjective but reflected a real change in brain activity.
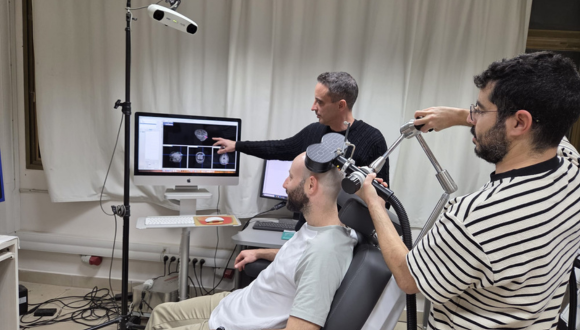
Illustration of the experimental setup
Special significance in the aftermath of October 7
These findings carry particular importance for IDF soldiers, members of the security forces, civilians exposed to the terror attacks of October 7, survivors of the massacre, and victims of shootings and abductions — Israeli populations in which the prevalence of PTSD is expected to be especially high. Many of them report experiencing intense intrusive memories months after the events. The potential development of a short, noninvasive treatment that directly targets the mechanisms underlying traumatic memories could become a valuable component of the national rehabilitation effort.
What Comes Next
According to the researchers, although this was a preliminary study conducted in a small group and did not include a control group, it provides clear proof of feasibility. Larger, controlled clinical trial is already underway at Tel Aviv University, and is required to assess the method’s effectiveness and long-term impact. If the findings are confirmed, this may represent a fundamental shift in the way traumatic memories are treated — addressing not only its emotional consequences, but the underlying neural root itself.
Prof. Nitzan Censor concludes: “These preliminary findings point to a conceptual shift in how we can approach the treatment of PTSD. We are attempting to intervene, in a targeted manner, in the brain mechanism of memory itself — at the moment when it ‘reopens’ and becomes amenable to change. The fact that we observed a consistent reduction in intrusive memories, alongside a measurable change in brain activity, is encouraging. It is important to emphasize that these are still very early results. Nevertheless, especially in light of the current reality in Israel, we hope that continued, comprehensive clinical research will eventually make it possible to develop a noninvasive and accessible treatment that will help many soldiers and civilians return to functional lives, free from the constant intrusion of traumatic memories.”


















.png)